- GST No. 27ACJPL6670Q1ZQ
- Send SMS
- Send Email
500 Kilogram (MOQ)
| Business Type | Manufacturer, Exporter |
| Country of Origin | India |
| Type | Drug Intermediate And Specialty Chemicals |
| Brand Name | AL-S-DPE |
| Click to view more | |
Preferred Buyer From
| Location | Worldwide |
Product Details
Color
Colorless To Yellow Liquid
CAS No.
2202-69-9
Other Names
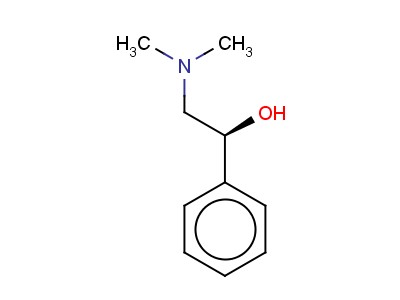
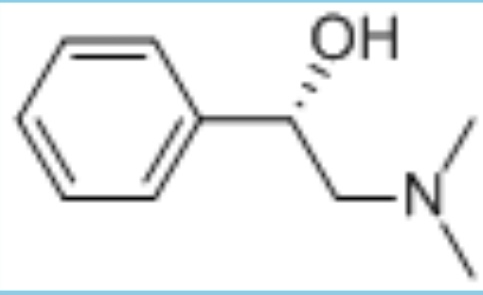
((S)-2-Dimethylamino-1-phenyletanol
MF
C10H15NO
Purity
96.5%
Name
AL-S-DPE
Product Code
AL-S-DPE
Port
Mumbai
Payment Terms
L/C, T/T
Delivery Time
45 days from PO
Packaging Details
Packed in HDPE Jerry can with export worthy packing and store below 8 deg.
This product manfucture in the state of art manfuafacturing facility with GMP equipvalence and itnernational standard facility. Is audited by many Indian and overseas compnay for their KSM for USFDA API. Is a light yellow viscuos liquid and should store below 8 deg. The facility is stricktly and stingently governed by current ICH Guidelines.
Looking for "((S)-2-Dimethylamino-1-Phenylethanol" ?
Kilogram



![6-methoxy-2-(4-methoxyphenyl)-benzo[b]thiophene 6-methoxy-2-(4-methoxyphenyl)-benzo[b]thiophene](https://2.wlimg.com/product_images/bc-small/2021/3/3984593/6-methoxy-2-4-methoxyphenyl-benzo-b-thiophene-1614591485-5650065.jpeg)


![7-BROMO-2,3-DICHLOROPYRIDO[2,3-B]PYRAZINE 7-BROMO-2,3-DICHLOROPYRIDO[2,3-B]PYRAZINE](https://2.wlimg.com/product_images/bc-small/2022/3/3984593/7-bromo-2-3-dichloropyrido-2-3-b-pyrazine-1647244258-6242843.jpeg)